Elekta has strengthened its global leadership in stereotactic radiosurgery with the announcement that its Leksell Gamma Knife® platform has now received CE mark approval for treating mesial temporal lobe epilepsy (MTLE) in adults. This new European regulatory clearance builds on Esprit’s existing U.S. FDA 510(k) clearance for refractory, drug‑resistant MTLE, expanding access to a non‑invasive therapeutic option for patients across markets.
With this CE marking, healthcare providers in regions recognizing EU regulatory standards can now offer Esprit as a precision alternative to open neurosurgery for carefully selected epilepsy cases. Esprit’s demonstrated submillimeter accuracy and highly targeted dose delivery make it uniquely suited to treat epileptogenic zones while minimizing exposure to healthy brain tissue, an essential advantage for patients seeking effective treatment with fewer side effects.
By expanding Esprit’s regulatory footprint, Elekta is accelerating access to a gentler, non‑invasive alternative for epilepsy patients whose seizures remain uncontrolled by medication and who may not qualify for conventional surgery. This milestone underscores the company’s commitment to delivering advanced technologies that protect both cognitive function and quality of life.
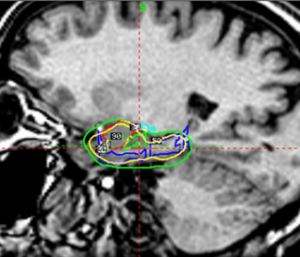
Read case study for Ruber International Hospital here.
